
The Bergeron Process explains precipitation growth in cold clouds. By cold clouds it is meant that the cloud contains ice particles or a mixture of liquid and ice particles. As with all clouds, water vapor moisture is present. Moisture becoming vapor from a liquid surface (evaporation) is easier than moisture becoming vapor from an ice surface (sublimation). Thus, saturation around a water surface requires more water vapor molecules than saturation around an ice surface. When an ice and water particle are present in a subfreezing environment, the vapor pressure will be less around the ice particle and greater around the liquid drop. This creates a vapor pressure gradient. Just like how air flows from high pressure toward low pressure, moisture flows from high vapor pressure toward lower vapor pressure. Since the ice has the lower vapor pressure, the water vapor moisture moves toward and deposits on the ice (deposition) in a saturated environment. Thus, in a mixture of ice and liquid drops within a cloud in a subfreezing environment, the ice will grow faster than the liquid drops due to the Bergeron process. 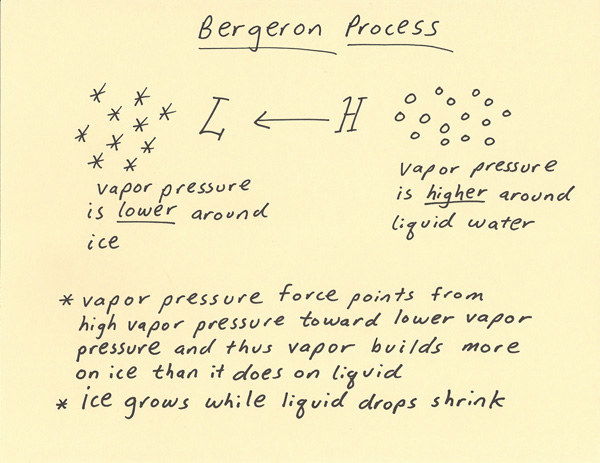 The dewpoint is the temperature at which saturation will occur over a liquid surface when the air is cooled. The frost point is the temperature at which saturation will occur over an ice surface when the air is cooled. The saturation point is different over a liquid surface (dewpoint) as compared to a frozen surface (frost point). The frost point is higher over a frozen surface and this results in deposition first occurring over the frozen surface when temperatures cool toward saturation. When considering the saturation point over water, the saturation point over ice is supersaturated as compared to that over water. This is another way of explaining how the ice grows while the water does not in a cold cloud. With respect to the dewpoint, the relative humidity is 100% surrounding the water but is greater than 100% surrounding the ice. Thus, the ice grows rapidly in order to try to bring the dewpoint back toward 100% around the ice. Since the vapor pressure gradient is continuously moving more water vapor toward the ice, this keeps the relative humidity (with respect to liquid water) higher than 100% over the ice. The reason this is important is that it explains rapid ice precipitation growth in cold clouds. Unlike liquid water on the ground, liquid water in the air can have a temperature of below freezing and still exist as water (called supercooled water). Supercooled water is very common in cold clouds. Thus, a cold cloud can have a temperature of -10 C for example and there will be a mixture of liquid and ice water. Once there is a mixture of liquid and ice then the Bergeron Process will quickly result in the cloud trending toward being all ice over time. This will not occur until there is some ice present. Temperatures typically need to drop well below freezing in a supercooled cloud before any of the liquid water has a chance to freeze. The colder the cloud gets then the more likely it is that some of the particles will freeze. Below around -40 C, any liquid water will spontaneously freeze in a cloud. At temperatures between freezing and -40 C , it is the Bergeron Process that will tend to change a greater percentage of cold cloud from supercooled water to ice particles once some ice particles are present. 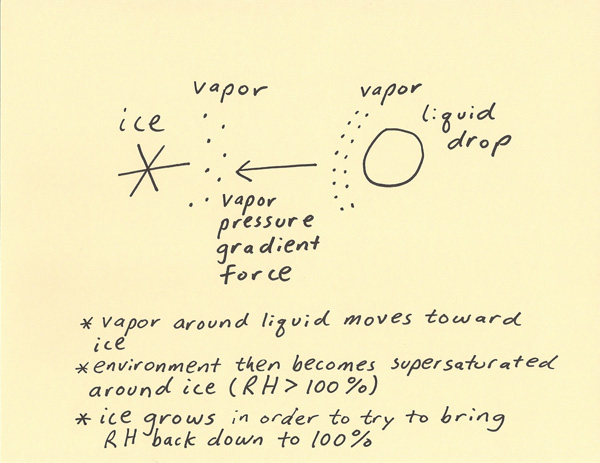 |